miQ3 Microstructure Database
miQ3 is an online resource providing the most accurate database of RLD microstructure to support generic development.
Diverse Product Library
miQ3 is an ever-growing database of RLD products providing all quality attributes critical to support regulatory approval without the need for clinical bioequivalence studies. New data is constantly being curated with a focus on supporting products where microstructure is critical to performance, and RLD's where patents are near expiry or those that have expired recently.
Inter-Sample Variability
miQ3 contains data regarding the variability observed between different individual doses of RLD products. This provides critical information necessary to an effective and efficient scale up of a potential generic.








Intra-Sample Microstructure
Containing data from ultra high-res microscopy techniques such as 3D FIB or synchrotron, miQ3 provides all the necessary critical quality attributes for characterization of RLD products. This offers critical insight significantly speeding up reverse engineering efforts. Additionally, it can be used to support regulatory filing by providing equivalence at the nanoscopic level.



Directly Compare Generics
All the microstructure characterization provided by miQ3 has been computed using the digiM I2S software platform. Containing numerous capabilities for characterizing drug formulation microstructures, miQ3 users can be confident they are making 1-to-1 comparisons between their in-house formulations and RLD products. Learn more about I2S here.
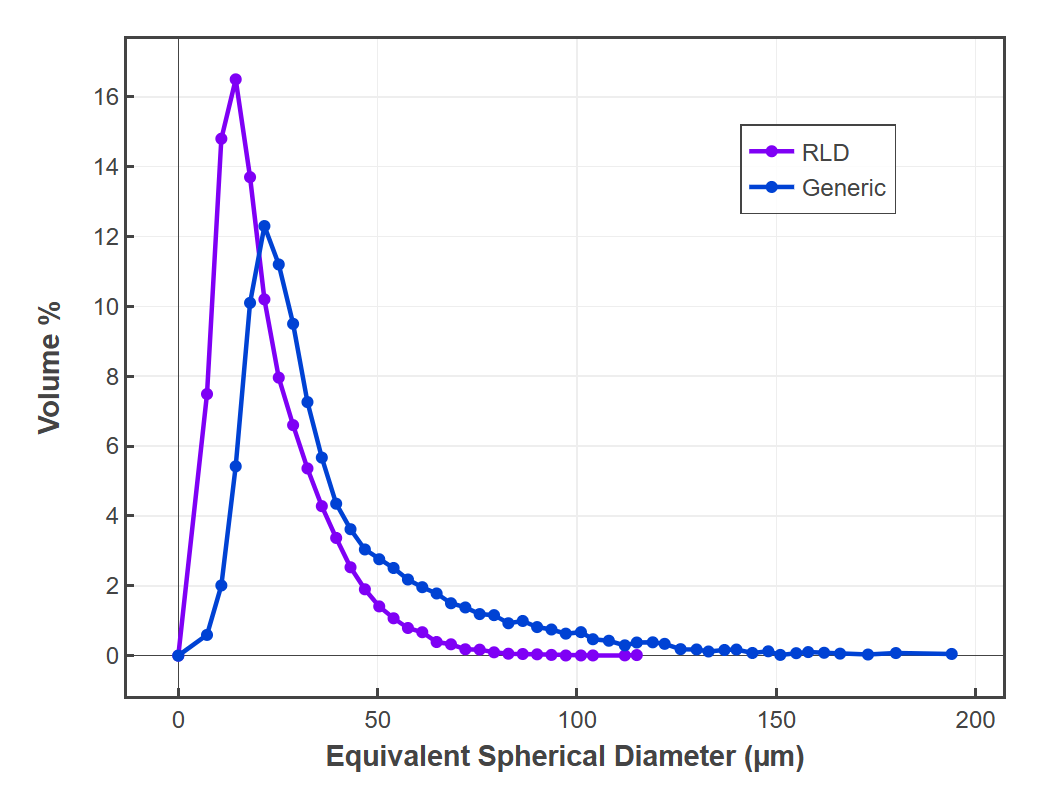
Release Performance Assessment
Through tight integration with the digiM software portfolio, miQ3 offers dissolution testing with validated methods for predicting in-vitro release in-silico. Using these models in dissoLab for your generic formulations, release can be compared with RLD products entirely in-silico. Additional software subscription may be required.









